Iron Anode Cathodic Protection Supplier & Manufacturer In China
Iron anodes are a cost-effective material in the field of cathodic protection. Wstitanium iron anodes, through unique alloy composition and manufacturing technology, achieve core advantages such as low consumption rate, uniform current distribution, and wide environmental adaptability. They have been gradually applied in key areas such as petrochemicals, marine engineering, and municipal construction.
- Sacrificial Iron Anodes
- ICCP Iron Anodes
- Pure Iron Anodes
- Cast Iron Anodes
- Iron-Silicon Anodes
- Iron-Nickel Anodes
- MMO Iron Anodes
- Custom-Made Iron Anode

Iron Anode and Cathode Protection Factory
Metal corrosion is a perpetual challenge in the industrial sector. Statistics show that the global economic losses due to metal corrosion annually account for 3%-5% of GDP, far exceeding the total losses from natural disasters. Among numerous anti-corrosion technologies, cathodic protection, due to its cost-effectiveness and long-term effectiveness, has become a core protection solution for large metal structures such as buried pipelines, offshore platforms, and storage tanks. Wstitanium, as a manufacturer of iron anodes, provides a comprehensive comparison of its performance with aluminum, zinc, magnesium anodes, and MMO titanium anodes, covering types, working principles, manufacturing techniques, and applications, offering you authoritative reference information.

Pure Iron Anode
Made from high-purity iron, typically with a purity of ≥99.5%, it is mainly used for temporary cathodic protection of small metal components. It is not suitable for long-term use.

High-Silicon Cast Iron Anode
Silicon content: 10%–14%. It exhibits excellent corrosion resistance and electrical conductivity, and is commonly used in strong electrolyte environments such as seawater and brine.

Nickel-Iron Anode
With a nickel content of 10% to 30%, it offers superior corrosion resistance compared to pure iron and is suitable for corrosive environments such as those involving chemical media and seawater.

Cast Iron Anode
With a carbon content of 2% to 4.3%, it is low in cost and high in hardness, making it suitable for cathodic protection in low-corrosive media such as soil and freshwater.

Iron Sacrificial Anode
Electrode potential of iron (-0.54 V, standard state) is lower than that of copper, titanium, etc., and it forms a galvanic cell with the protected metal in an electrolyte environment (seawater, soil, wastewater).

ICCP Ferrosilicon Anode
Iron anodes, acting as auxiliary anodes, provide electrons through an external power supply and are suitable for large storage tanks, long-distance pipelines, and offshore platforms.
Working Principle of Iron Anodes
The core of metal corrosion is an oxidation-reduction reaction. Steel in a humid environment spontaneously forms a corrosion cell: oxidation of iron occurs at the anode. Iron atoms lose electrons to form Fe²⁺, with the reaction equation: 2Fe → 2Fe²⁺ + 4e⁻. At the cathode region, oxygen reduction occurs, where oxygen combines with electrons and water to form OH⁻, with the reaction equation: O₂ + 4e⁻ + 2H₂O → 4OH⁻; Fe²⁺ further combines with OH⁻ to form ferrous hydroxide (Fe(OH)₂), which is gradually oxidized into rust (Fe₂O₃・nH₂O), leading to continuous damage to the steel structure.
The formation of a corrosion cell requires three conditions: metals with different potentials (or different regions of the same metal), an electrolyte environment (such as soil, seawater, rainwater), and a metallic pathway. The core of cathodic protection is to disrupt the formation of the corrosion cell through external intervention, making the protected metal as a whole the cathode.
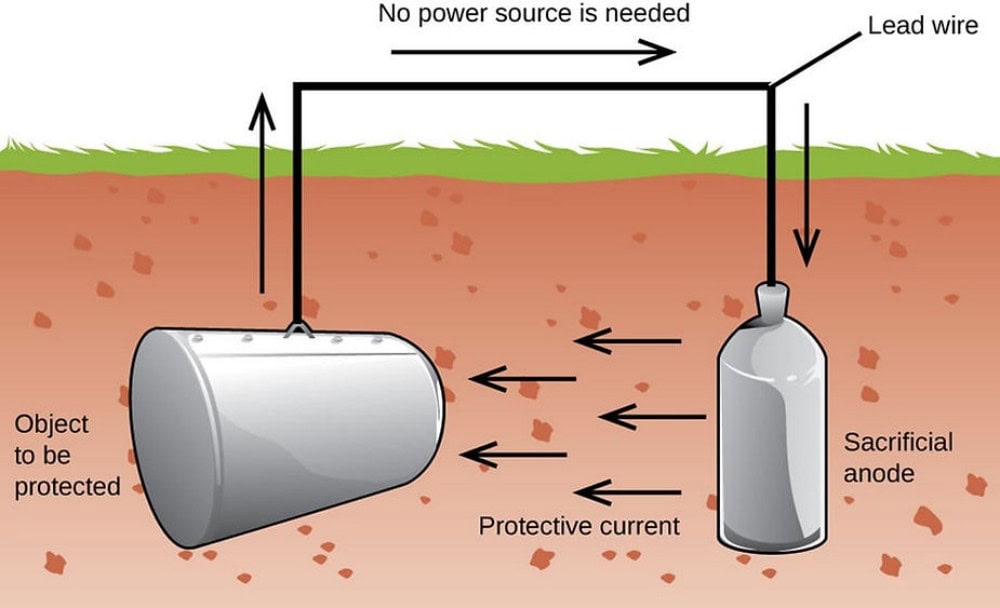
Sacrificial Protection
The electrode potential of the iron anode is lower than the self-corrosion potential of steel. When the two are connected by a wire and are in the same electrolyte environment, a spontaneous galvanic cell is formed. The iron anode acts as the anode, undergoing oxidation (Fe → Fe²⁺ + 2e⁻), and the released electrons flow through the metallic pathway to the protected steel structure, shifting its surface potential negatively to a range where corrosion stops (usually -0.85V to -1.2V vs SCE). At this point, the oxidation reaction on the steel surface is inhibited, and all anodic reactions are concentrated on the iron anode, achieving protection of the steel structure through the sacrifice of the anode.
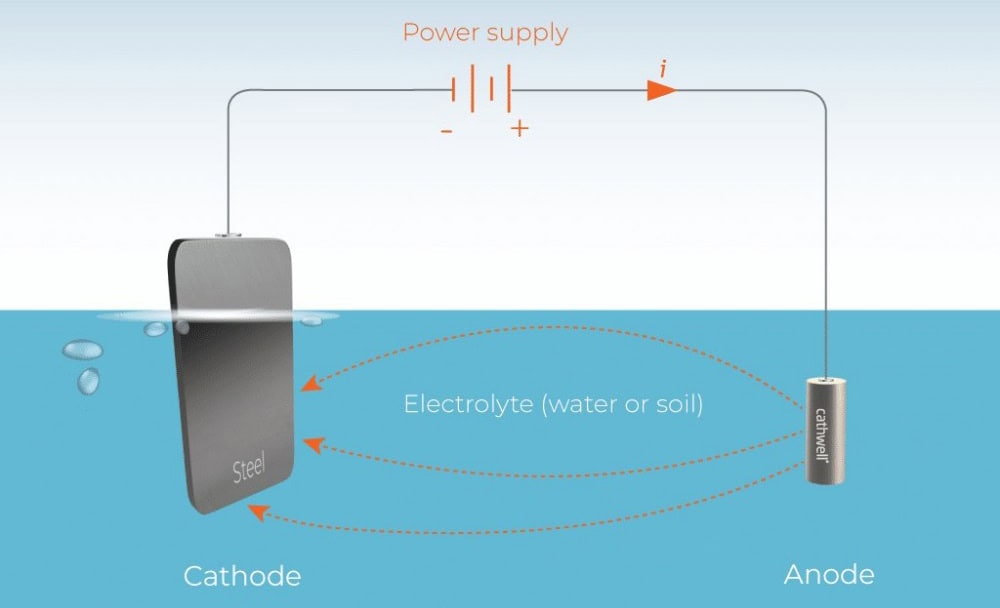
Impressed Current Protection
In this system, the iron anode is connected to the positive terminal of an external DC power supply via a wire, and the protected metal is connected to the negative terminal. After power is applied, the external power supply forces electrons to flow from the iron anode to the protected metal, providing the protected metal surface with sufficient electrons to inhibit the oxidation of Fe. Oxygen or chlorine evolution reactions occur on the anode surface (e.g., in a chlorine-containing medium: 2Cl⁻ → Cl₂ + 2e⁻), maintaining the stability of the current loop. Precise control of the potential of the protected metal achieves long-term stable corrosion protection.
Iron Anodes vs. Other Anodes
Iron anodes differ significantly from aluminum anodes, zinc anodes, magnesium anodes (the main types of sacrificial anodes), and MMO titanium anodes (the main type of impressed current anodes) in terms of electrochemical performance, applicable environments, and cost.
| Performance | Iron Anode | Aluminum Anode | Zinc Anode | Magnesium Anode | MMO Titanium Anode |
| Electrode Potential (vs SCE) | -0.44 ~ -0.95V | -1.05 ~ -1.10V | -1.10 ~ -1.15V | -1.50 ~ -2.00V | Inert (no corrosion) |
| Potential Difference with Steel | 0.3 ~ 0.5V | 0.8 ~ 1.2V | 0.2 ~ 0.5V | 1.5 ~ 2.0V | Depends on external POWER supply |
| Current Efficiency | 80%~85% | 80%~90% | 85%~95% | 50%~70% | 95%~98% |
| Theoretical Capacity (Ah/kg) | 1200~1500 | 2980 | 820 | 2200 | – (no consumption) |
| Actual Capacity (Ah/kg) | 1000~1200 | 2000~2500 | 700~750 | 1400~1800 | – (no consumption) |
| Annual Consumption Rate (kg/(A.a)) | 0.5~0.8 | 0.3~0.5 | 0.6~0.9 | 1.2~1.8 | Negligible |
| Polarization Rate (mV/A) | 40~60 | 30~50 | 20~40 | 50~80 | 10~30 |
Iron Anode Applications
Iron anodes operate stably in soil (resistivity 5-100 Ω·m), seawater, freshwater, and weakly acidic/alkaline chemical media. They are particularly suitable for medium-resistivity environments and long-term protection scenarios, such as buried pipelines, storage tank bottoms, and auxiliary protection for offshore platforms. Thanks to their excellent overall performance, iron anodes have been widely used in various fields, including petrochemicals, marine engineering, municipal construction, and energy and power.
Petrochemical Industry
In long-distance oil and gas pipelines, iron anodes are spaced 50-100m apart and buried along both sides of the pipeline. A stable protective current is provided by an impressed current system, maintaining the pipeline protection potential at -0.85V to -1.0V (vs CSE), reducing the corrosion rate by more than 90%.
Storage Tank Bottom Plate Protection: The bottom plates of large crude oil and chemical storage tanks are susceptible to soil corrosion. A mesh anode is used for comprehensive protection. The anode mesh is spaced 5-10cm from the tank bottom plate, powered by a sacrificial anode system or an impressed current system, ensuring a protection potential uniformity error of <5%.

Chemical Equipment Corrosion Protection: In chemical reactors, heat exchangers, pipelines, and other equipment, plate anodes are used for cathodic protection, suitable for weakly acidic and weakly alkaline media environments. The anodes are fixed to the inner wall of the equipment with bolts and used in conjunction with an external power supply, ensuring stable operation of the equipment under conditions of 80℃ and pH 4-10.
Marine Engineering
Offshore platform steel piles, constantly immersed in seawater, face serious corrosion and marine biofouling problems. Tubular anodes are fixed around the steel piles at a depth of 5-10m, providing a long-term stable protective current.
Ships and Ports: Plate-shaped sacrificial anodes are used to protect ship ballast tanks and hulls. Iron anodes are welded to the ship’s structure, forming a galvanic cell with the hull. In the seawater environment, the anode slowly corrodes, releasing a protective current that shifts the hull potential to below -0.9V, effectively preventing seawater corrosion and pitting.

Municipal Construction
Urban buried gas pipelines traverse different soil environments. Iron anodes are buried at pipeline intersections, valve wells, and other critical locations. A remote-controlled impressed current system ensures that the protective potential of the pipeline meets the required standards (≥95%) in complex soil environments.
Wastewater Treatment Facilities: Applied to metal structures in wastewater treatment plants, such as reaction tanks, sedimentation tanks, and sludge pipelines. Iron anodes are suitable for the weakly acidic, high-chloride ion environment of wastewater. The anodes are installed on the inner wall of the structure and used in conjunction with an external power supply, with a current density controlled at 50-100mA/m², effectively preventing structural damage caused by wastewater corrosion.

Bridges and Tunnels: Steel piles and steel box girders of cross-sea bridges and underwater tunnels use a combined protection scheme of iron anodes and coatings. The anodes are plate-shaped or tubular and installed at critical corrosion points of the steel structure. An impressed current system provides the protective current, working synergistically with the anti-corrosion coating to extend the service life of the steel structure to over 100 years.
Conclusion
Iron anodes serve as a core material in cathodic protection technology. Their working principle is based on the electrochemical corrosion inhibition mechanism, which shifts the potential of the protected metal surface to a safe range through the spontaneous current of a sacrificial anode or the forced current of an external power supply system, thereby preventing corrosion.
Compared to aluminum, zinc, and magnesium sacrificial anodes, iron anodes offer significant cost-effectiveness advantages in scenarios requiring moderate service life; compared to MMO titanium anodes, they have lower initial investment costs. Iron anodes have been widely used in petrochemicals, marine engineering, municipal construction, and power generation, providing a reliable solution for corrosion protection.
