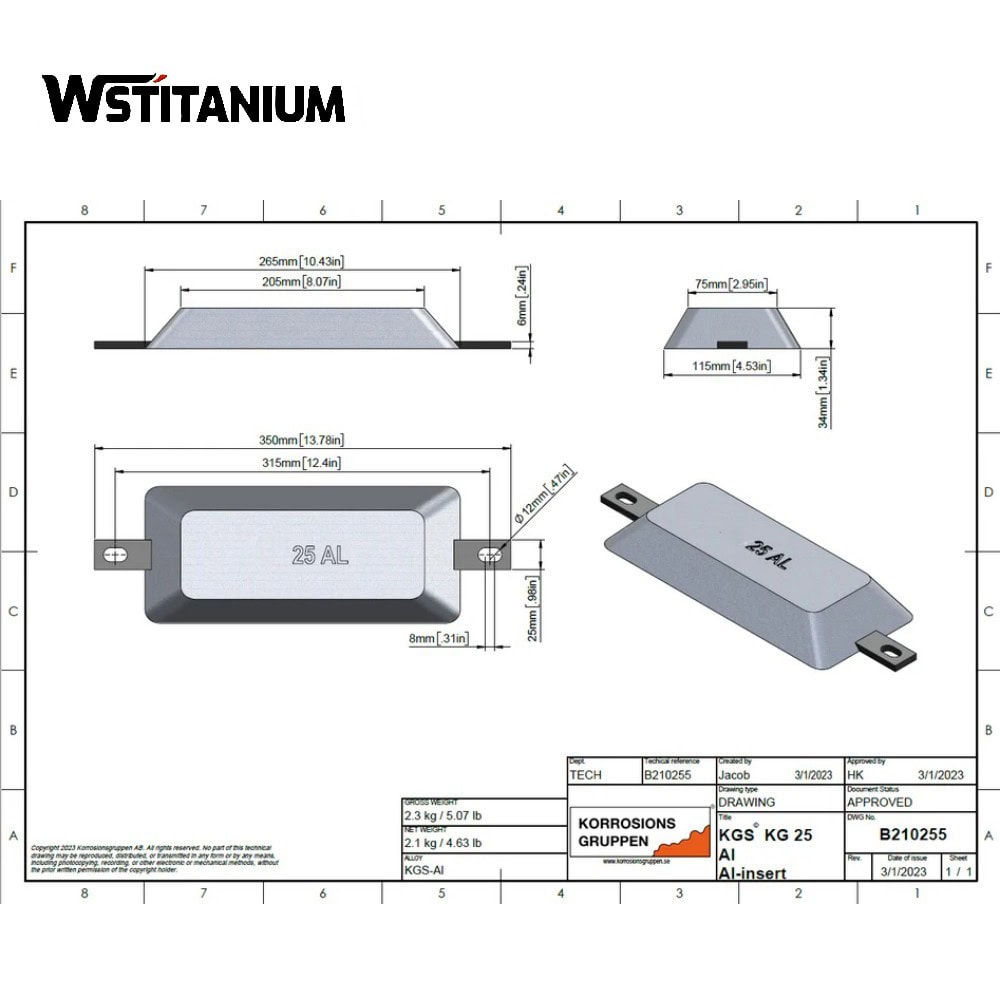
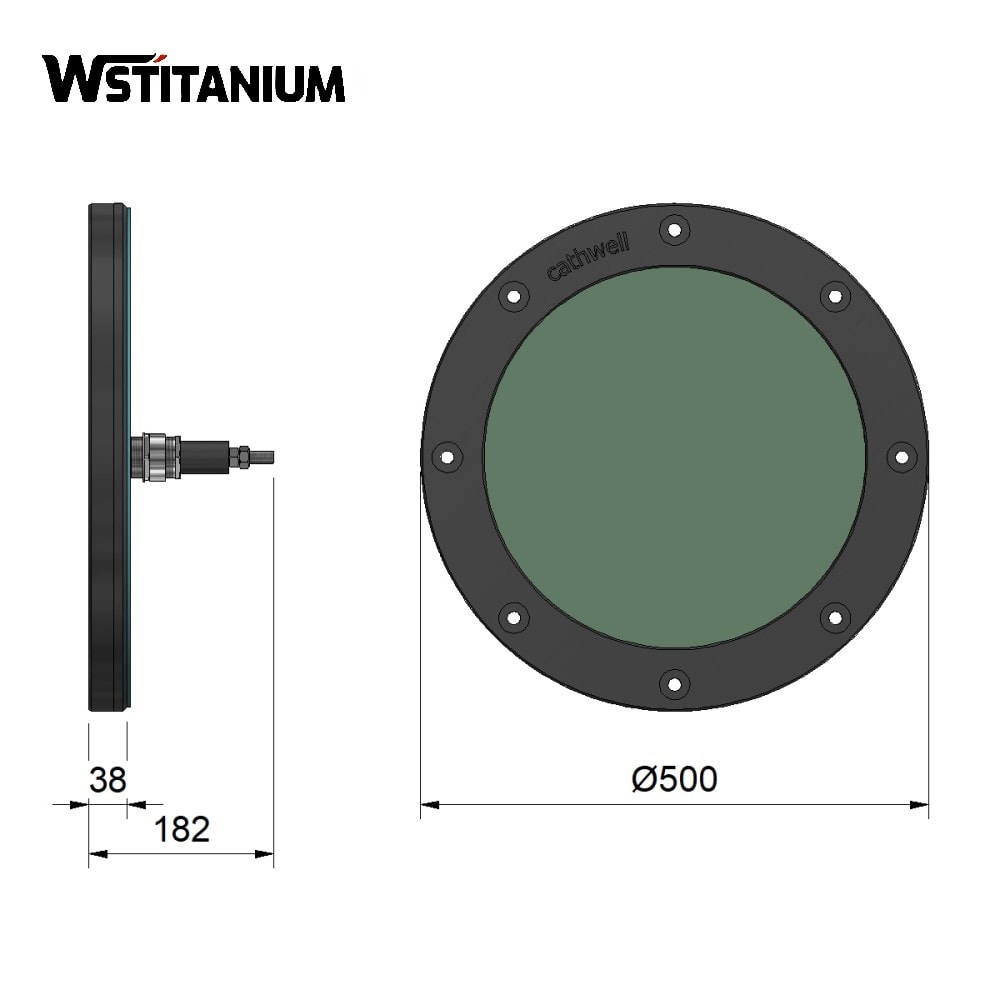
Hull anode
Wstitanium’s hull anodes are heavier than those of competitors and offer superior cathodic protection performance. Zinc anodes, aluminum anodes, and magnesium anodes are used to prevent corrosion in underwater hull sections, ballast tanks, drilling water tanks, and cargo holds. These sacrificial anodes meet the standards specified in Mil-Spec A-18001, Mil-Spec A-18001A, and Mil-Spec A-18001K.
- MMO Anodes
- Zinc Sacrificial Anodes
- Aluminum Sacrificial Anodes
- Magnesium Sacrificial Anodes
Wstitanium Workshop
Our Powerful Facilities

Ship Hull Anode Manufacturer in China
Highly corrosive, high-salinity, and high-humidity seawater can cause severe electrochemical corrosion to ship hull plates, propellers, rudders, and seabed valve boxes. The ship hull anode, as a core component of the ship’s cathodic protection system, effectively inhibits metal corrosion reactions by sacrificing itself or through the application of an external current. Wstitanium, a trusted manufacturer of cathodic protection hull anodes in China, develops technologies categorized into two main types: sacrificial anode cathodic protection and impressed current cathodic protection.
Hull Anode Type
Based on the different principles of cathodic protection, ship hull anodes are divided into two main categories: sacrificial anodes and impressed current anodes. Sacrificial anodes are the most widely used in civilian vessels due to their lack of need for an external power source, ease of installation, and low maintenance costs; impressed current anodes are suitable for long-term corrosion protection of large, specialized vessels and offshore engineering equipment.

Aluminum Sacrificial Anode
Seawater, brackish water, low resistivity waters; merchant ships, fishing boats, coastal ships.

Zinc Sacrificial Anode
High-salinity seawater; ocean-going ships, deep-sea operation platforms.

Magnesium Sacrificial Anode
Fresh water, high-resistivity inland waters; inland river ships, lake operation ships.
Hull Sacrificial Anode Elements
The composition design of sacrificial anodes must meet the requirements of having a potential more negative than the ship’s carbon steel, high current efficiency, uniform corrosion, and resistance to passivation. Different types of sacrificial anodes have clear industry standards regarding alloy element ratios and specifications (e.g., GB/T 4948-2002 “Zinc-Aluminum-Cadmium Alloy Sacrificial Anodes,” GB/T 4950-2002 “Zinc-Aluminum-Magnesium-Copper Alloy Sacrificial Anodes,” GB/T 17848-2018 “Aluminum-Based Sacrificial Anodes”).
| Sacrificial Anode | Element | Impurity Element Limits | Electrochemistry | L × W × H/mm |
| Zinc-Aluminum-Magnesium-Copper | Zn: Balance; Al: 1.0%~1.5%; Mg: 0.08%~0.12%; Cu: 0.05%~0.10% | Fe: 0.005%; Si: 0.01%; | Open circuit potential: -1.08~-1.15V; Current efficiency: ≥80%; Actual capacitance: ≥820A·h/kg. | 400×150×80; 550×220×110; 700×280×140. |
| Aluminum-Zinc-Indium | Al: Balance; Zn: 4.0%~5.0%; In: 0.02%~0.05% | Fe: 0.10%; Cu: 0.01%; Si: 0.10% | Open circuit potential: -1.18~-1.25V; Current efficiency: ≥90%; Actual capacitance: ≥2600A·h/kg. | 600×200×100; 750×250×120; 900×300×150. |
| Aluminum-Zinc-Indium-Tin | Al: Balance; Zn: 4.5%~5.5%; In: 0.03%~0.06%; Sn: 0.02%~0.04% | Fe: 0.08%; Cu: 0.008%; Si: 0.08% | Open circuit potential: -1.20~-1.28V; Current efficiency: ≥90%; Actual capacitance: ≥2600A·h/kg. | 500×180×90; 650×230×110; 850×290×140. |
| Magnesium-Aluminum-Zinc-Manganese | Mg: Balance; Al: 5.3%~6.7%; Zn: 2.5%~3.5%; Mn: 0.15%~0.60% | Fe: 0.005%; Cu: 0.005%; Ni: 0.003% | Open circuit potential: -1.55~-1.60V; Current efficiency: ≥50%; Actual capacitance: ≥1200A·h/kg. | 300×120×60; 450×180×90; 600×240×120. |
Sacrificial Anode Gallery
Elements of ICCP Anodes
The core requirements for impressed current anodes are high conductivity, strong corrosion resistance, high current density, and long lifespan. Their performance is directly determined by the base material and coating material. Currently, mixed metal oxide (MMO) anodes are the most popular product. Graphite anodes and lead-silver alloy anodes are gradually being replaced.
| ICCP Anode | Substrate | Coating | Performance | Specifications | Design Life |
| Ruthenium-Iridium MMO Anode | Pure Titanium (Gr1/Gr2) | RuO₂: 20%~30%; IrO₂: 70%~80%; Coating Thickness: 5~10μm | Operating current density: 100~200A/m²; Polarization rate: ≤5mV/year; Corrosion resistance: No obvious loss after 10 years immersion in seawater. | Strip: Φ20×1000~3000mm; | 10~20 years |
| Plate: 200×500×3mm; | |||||
| Mesh: 500×500×2mm | |||||
| Iridium-Tantalum MMO Anode | Pure Titanium (Gr1/Gr2) | IrO₂: 30%~40%; Ta₂O₅: 60%~70%; Coating Thickness: 8~12μm | Operating current density: 150~250A/m²; Strong corrosion resistance; Suitable for high salinity and strong oxidizing environments. | Tube: Φ25×1500~4000mm; | 15~25 years |
| Plate: 300×600×4mm |
Hull ICCP MMO Anode Gallery
Working Principle of Sacrificial Anodes
Sacrificial anode cathodic protection is based on the galvanic cell principle of electrochemical corrosion. The main component of a ship’s hull steel plate is carbon steel. In an electrolyte environment such as seawater, countless tiny galvanic cells are formed on the surface of the carbon steel. The area with a more positive potential acts as the cathode, and the area with a more negative potential acts as the anode. Oxidation (corrosion) occurs in the anodic region, while the cathodic region remains stable.
The standard electrode potential of the sacrificial anode material (zinc, aluminum, magnesium) is much lower than that of the ship’s carbon steel. When the sacrificial anode is connected to the ship’s steel plate via a wire or directly, and both are immersed in seawater, they form a new macroscopic galvanic cell.
In this case, the sacrificial anode acts as the anode (negative electrode) of the galvanic cell, and the ship’s steel plate acts as the cathode (positive electrode). Under the action of the electrolyte, the sacrificial anode preferentially undergoes oxidation, releasing electrons and metal ions:
- Zinc anodizing reaction: Zn→Zn 2+ +2e −
- Aluminum anodizing reaction: Al→Al 3+ +3e −
- Magnesium anodizing reaction: Mg→Mg 2+ +2e −
The released electrons flow through seawater or connecting wires to the ship’s steel plates, causing a large accumulation of electrons on the surface of the steel plates, thereby inhibiting the oxidation reaction of the carbon steel itself (Fe→Fe2++2e−).
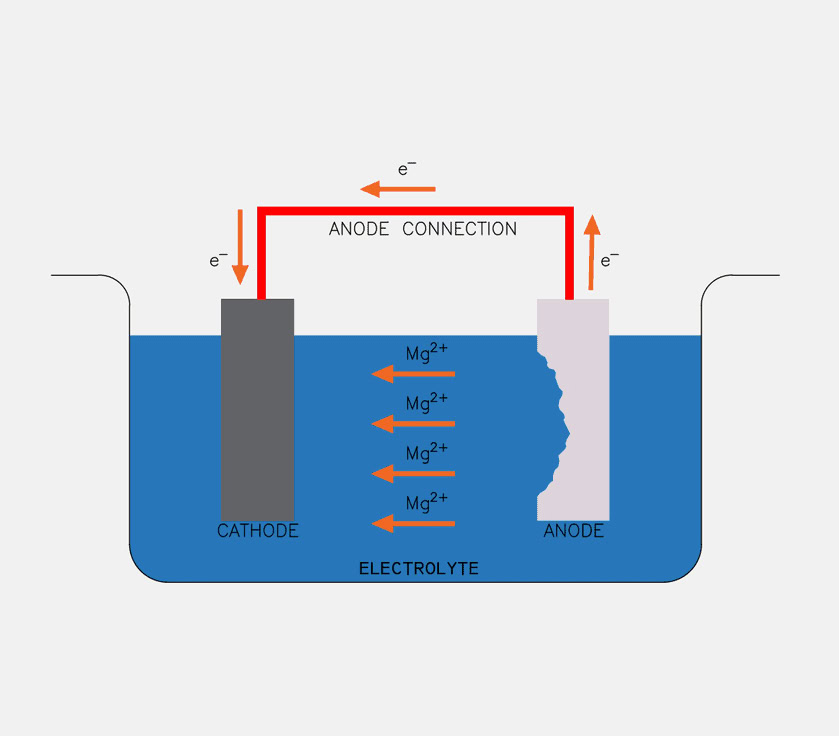
Meanwhile, dissolved oxygen in seawater gains electrons on the surface of the ship’s steel plates, undergoing a reduction reaction: (O2+2H2O+4e−→4OH−). The sacrificial anode provides a protective current to the ship’s steel plates through its continuous corrosion and consumption, until the anode is completely depleted. Sacrificial anodes do not require an external power source, and the magnitude of the protective current can be automatically adjusted through the number, size, and distribution of the anodes, making them suitable for corrosion protection in most civilian vessels.
Installation Locations of Hull Anodes
The risk of corrosion varies in different parts of the hull. The installation locations of the anodes must be precisely determined based on corrosion intensity, water flow velocity, and structural characteristics to ensure that the protective current covers all metal components of the hull. The specific installation locations and protection targets are shown in the table below:
| Installation Location | Type | Anode Shape | Protection Target | Design Basis |
| Ship Bottom Flat Area | Zn/Al Sacrificial Anode | Block, Plate | Prevent uniform corrosion and pitting of the ship bottom steel plate. | Calculated by the hull bottom area: 1~2 pieces of 50kg-grade anodes shall be arranged per 10 m2. |
| Ship Hull Side | Zn/Al Sacrificial Anode | Strip | Protect the hull side steel plate and resist corrosion from sea wave impact. | 1 strip anode shall be arranged every 2~3 m along the longitudinal direction of the hull side. |
| Propeller & Tail Shaft | Zn/Al Sacrificial Anode | Ring, Strip | Prevent propeller cavitation corrosion and tail shaft electrochemical corrosion. | 1 ring anode installed on each side of the propeller; 2~3 strip anodes arranged at the tail shaft sleeve. |
| Rudder Blade & Rudder Stock | Zn/Al Sacrificial Anode | Block, Strip | Protect the rudder blade surface and rudder stock connection parts. | 2 block anodes installed on each side of the rudder blade; 1~2 strip anodes arranged near the rudder stock. |
| Ballast Tank Inner Wall | Zn Sacrificial Anode / MMO Anode | Block, Tube | Resist corrosion in the alternating dry-wet environment inside the ballast tank. | Arranged by tank capacity: 1 piece of 30kg-grade anode installed per 50 m糧 of tank volume. |
| Sea Chest & Sea Water Pipeline | Al Sacrificial Anode / MMO Anode | Rod, Tube | Prevent local corrosion of the valve box inner wall and pipelines. | 2~3 rod anodes installed inside the valve box; 1 tube anode installed every 5~10 m in the pipeline. |
| Bottom of Large Ocean-Going Vessels | MMO Anode | Mesh, Plate | Provide long-term protection for long-distance ocean navigation. | Evenly arranged by hull bottom area, with current density controlled at 150~200 A/m2. |
Anode Installation Precautions
Before installing the anode, the paint, rust, and oil stains on the hull installation area must be cleaned to ensure good contact between the anode and the hull steel plate, preventing excessive contact resistance from affecting the protective current transmission. For impressed current anodes, an insulating gasket (such as rubber or nylon gasket) must be installed between the anode and the hull to prevent short circuits. Sacrificial anodes are generally secured with bolts. The bolts should be made of corrosion-resistant materials (such as stainless steel or titanium), and anti-corrosion grease should be applied to the bolt connections. Impressed current anodes must be fixed with a special bracket to ensure secure installation and resistance to wave impact.

After installation, a reference electrode should be used to measure the potential distribution on the hull surface to ensure that the potential value is maintained at -0.85~-1.20V (relative to a saturated calomel electrode) without any unprotected areas. The antifouling paint in the anode installation area should be of a type that does not contain copper ions to prevent copper ions from reacting with the anode to form a passivation film, which would reduce anode activity.
Selection of Hull Anodes
The type of anode should be selected based on the resistivity, salinity, and temperature of the water in which the vessel operates. Aluminum sacrificial anodes are preferred for high-salinity seawater. Zinc-based anodes are suitable for brackish water, and magnesium-based anodes for freshwater. Sacrificial anodes are typically used for small vessels, while impressed current anodes are preferred for large ocean-going vessels and special-purpose ships.
The anode with the best cost-effectiveness should be selected, taking into account procurement costs, installation costs, maintenance costs, and service life. The anode material must be compatible with the hull metal, antifouling paint, and seawater piping to avoid chemical reactions that could lead to performance degradation.

Calculating the anode requirements: Based on the metal surface area of the hull requiring protection, the protective current density, and the anode capacity, calculate the number and specifications of the anodes. The calculation formula is:
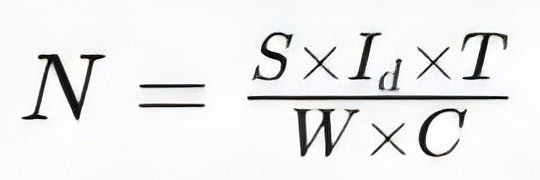
(Where N is the number of anodes, S is the protected area, Id is the protective current density, T is the design life, W is the weight of a single anode, and C is the actual capacity of the anode.)
Typical Hull Anode Applications
| Vessel Type | Navigation Environment | Corrosion Risk | Recommended Anode | Shape | Life (Years) |
| 5000 DWT Cargo Ship | Brackish Water (Salinity 10‰~20‰) | Medium | Zn-Cu-Mg-Cu Anode | Block + Strip | 3~5 |
| 30,000 DWT Ocean Vessel | High Salinity Seawater (Salinity 30‰~35‰) | High | Al-Zn-Cu-Sn Anode + MMO Anode | Plate + Mesh | 6~8 |
| 500 DWT Inland Ship | Fresh Water (Resistivity >10000Ω·cm) | Low | Mg-Al-Zn-Mn Anode | Rod + Block | 2~3 |
| LNG Carrier | High Salinity Seawater + Low Temperature Environment | Extremely High | Ir-Ta MMO Anode | Mesh + Tube | 15~20 |
| Small Fishing Vessel | Coastal Seawater (Salinity 25‰~30‰) | Medium | Zn-Al-Cd Anode | Block + | 3~5 |
Conclusion
Hull anodes are a core component of ship corrosion protection. Their selection, installation, and maintenance directly determine the safety and operating costs of the vessel. This guide systematically reviews the two main types of hull anodes (sacrificial anodes and impressed current anodes), detailing their elemental composition, specifications, shape design, working principles, installation locations, and selection methods.
Sacrificial anodes, due to their simple installation and low maintenance costs, are widely used in civilian vessels. Zinc-based, aluminum-based, and magnesium-based sacrificial anodes are suitable for brackish water, high-salinity seawater, and freshwater environments, respectively. Impressed current anodes, with their long-lasting protection and high current density, are suitable for large ocean-going vessels, LNG carriers, and other specialized ships. In practical applications, it is necessary to follow three main principles: environmental suitability, ship type suitability, and economic efficiency. The type and specifications of the anode should be precisely selected based on sailing area parameters, hull corrosion risk, and service requirements.