MGPS Anode for Cruise Ships
Certified: CE & SGS & ROHS
Shape: Requested
Diameter: Customized
Drawings: STEP, IGS , X_T, PDF
Shipping: DHL, Fedex, or UPS & Ocean Freight

20+ YEARS EXPERIENCE SENIOR BUSINESS MANAGER
Ask Michin For What You Want?
Marine organisms such as algae, shellfish, and barnacles, which are widespread in the marine environment, easily attach and grow inside the seawater pipes, condensers, and coolers of cruise ships, causing biofouling. The hazards of biofouling to large cruise ships are twofold: firstly, the attachment of marine organisms reduces the cross-sectional area of pipes and increases water flow resistance, causing a decrease in the heat exchange efficiency of the cooling system by more than 30%, leading to problems such as engine overheating and air conditioning malfunctions; secondly, acidic substances produced by marine organism metabolism accelerate pipe corrosion, shorten their lifespan, and increase the risk of leaks.
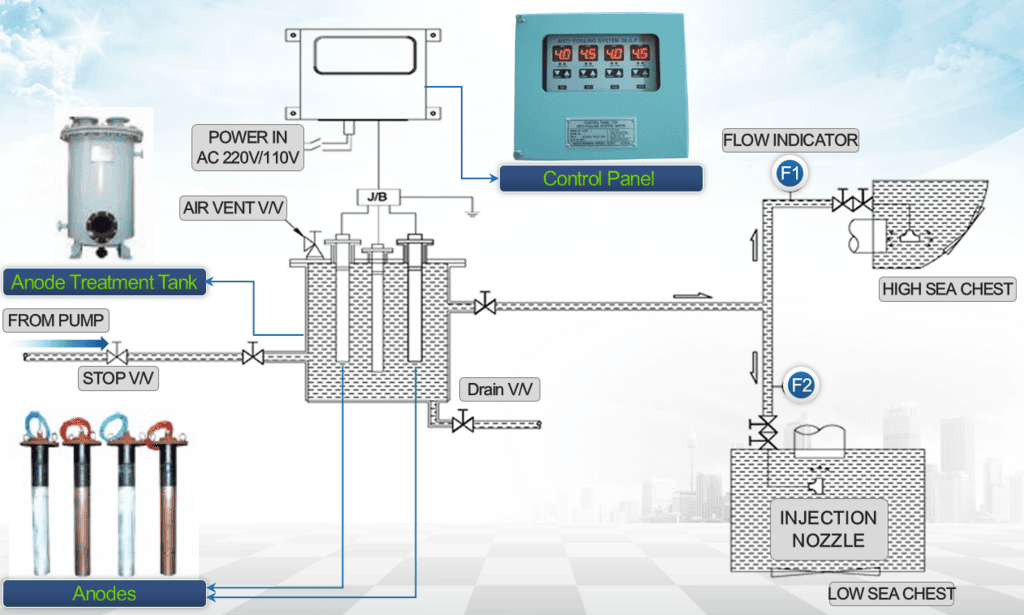
The Marine Growth Prevention System (MGPS) is a core technology for solving the problem of marine biofouling. It uses anodic electrolysis to generate specific ions, achieving dual protection against biofouling and corrosion.
MGPS Anodes
The type of MGPS anode needs to be precisely selected based on factors such as the sailing area of the large cruise ship, the pipe material, and the seawater system flow rate. Its core functions revolve around “inhibiting marine organism growth” and “protecting pipes from corrosion.”
Copper Anodes (Core Antifouling Anode)
Copper anodes are the core component in the MGPS system for inhibiting marine organism growth. They are primarily made of high-purity electrolytic copper (purity ≥99.95%). Some high-end products may add small amounts of zinc and tin alloys to improve anode dissolution uniformity. Copper anodes come in various specifications, with diameters mainly concentrated at 70mm, 82.5mm, 100mm, and 120mm, and lengths extending from 250mm to 600mm. For some special pipe systems, non-standard products with diameters of 3.5 inches to 5 inches and lengths of 12 inches to 36 inches can be customized.
The core advantage of copper anodes is their stable antifouling effect. The concentration of copper ions generated by electrolysis only needs to be maintained at 2 ppb (parts per billion) to effectively inhibit the attachment and growth of larvae of marine organisms such as algae, barnacles, and mussels. It is primarily suitable for tropical and subtropical waters with high biological activity, making it the preferred antifouling anode for large cruise ships navigating transoceanic voyages.
Aluminum Anode (Core Antifouling Anode)
The aluminum anode uses high-purity aluminum as the base material, with added alloying elements such as zinc, indium, and magnesium (e.g., Al-Zn-In alloy). Its main function is to provide corrosion protection for the steel seawater pipelines of cruise ships, while also enhancing the antifouling effect. Its specifications are basically matched with the copper anode, and can be flexibly selected according to the pipeline diameter and current requirements. Common diameters are 70mm-120mm, and lengths are 250mm-500mm.
The core function of aluminum anodes is to generate aluminum hydroxide flocculents through electrolysis. On one hand, this flocculent material adsorbs and kills marine larvae, enhancing the antifouling efficiency of copper anodes. On the other hand, aluminum hydroxide accumulates on the inner walls of steel pipes, forming a dense and stable protective film that isolates seawater from direct contact with the metal surface, inhibiting electrochemical corrosion.
Compared to zinc anodes, aluminum anodes have a higher theoretical capacitance (approximately 2800 Ah/kg), are lighter, and are more suitable for the lightweight requirements of large cruise ships. Furthermore, they offer more stable corrosion protection in high-salinity, high-velocity seawater environments. They are suitable for temperate, low-biological-activity sea areas, or for steel-based seawater cooling and fire protection piping systems on cruise ships.
Iron Anodes (Special Anti-corrosion Anodes)
Iron anodes, also known as ferrous anodes, are primarily made of soft iron and are mainly used to protect special material pipes in large cruise ships, especially copper-nickel alloy pipes (commonly found in high-temperature cooling systems and domestic water cooling pipes on cruise ships). Its specifications are relatively small, typically 50mm-80mm in diameter and 200mm-300mm in length, allowing for direct installation in small filters or pipe bends.
The core function of the iron anode is to generate ferrous ions through electrolysis, forming a stable protective oxide film on the inner wall of the copper-nickel alloy pipe. This passivation on the pipe surface inhibits pitting and crevice corrosion of the copper-nickel alloy by seawater. Unlike aluminum anodes, iron anodes do not have auxiliary antifouling functions and are solely focused on corrosion protection for specialized pipelines.
Working Principle of MGPS Anodes
The working principle of MGPS anodes on large cruise ships is based on electrolysis. Its core is the output of a stable, low-intensity direct current from the MGPS control cabinet, causing the anode to undergo an oxidation reaction in seawater. The released ions form a current loop with the cathode in the system, achieving dual protection against biofouling and pipe corrosion. The electrolysis reaction of MGPS anodes mainly consists of anodic oxidation and cathodic reduction reactions. Different anode materials produce different oxidation reaction products, resulting in varying protective functions.
Electrolysis Reaction of Copper Anodes
Anodic Oxidation Reaction: Under the action of direct current, the copper anode undergoes oxidation and dissolution, releasing copper ions. The reaction formula is: Cu → Cu²⁺ + 2e⁻.
Cathode Reduction Reaction: The iron cathode in the system (or the cruise ship’s metal pipes themselves) undergoes a reduction reaction. Water molecules in the seawater gain electrons, generating hydrogen gas and hydroxide ions. The reaction formula is: 3H₂O + 2e⁻ → H₂↑ + 2OH⁻.
Antifouling Mechanism: Copper ions (Cu²⁺) combine with hydroxide ions (OH⁻) generated by the cathode reaction to form cuprous oxide (Cu₂O) colloid. Cuprous oxide is highly toxic and can destroy the cell structure of marine larvae, inhibiting their attachment and growth. Simultaneously, copper ions flow with seawater through the pipes, covering the inner wall and forming a “toxic ion membrane,” which prevents marine organisms from proliferating within the pipes over a long period.
Electrolysis Reactions at the Aluminum Anode
Anodic Oxidation Reaction: Under direct current, the aluminum anode undergoes oxidation and dissolution, releasing aluminum ions. The reaction is: Al → Al³⁺ + 3e⁻.
Cathode Reduction Reaction: Consistent with the cathode reaction at the copper anode, hydrogen gas and hydroxide ions are generated. The reaction is: 3H₂O + 3e⁻ → 3/2H₂↑ + 3OH⁻.
Anti-corrosion mechanism: Aluminum ions (Al³⁺) combine with hydroxide ions (OH⁻) to form aluminum hydroxide (Al(OH)₃) flocculent precipitate. As seawater flows, this precipitate gradually adsorbs onto the inner wall of the steel pipe, forming a dense protective film that isolates the metal surface from electrochemical corrosion by seawater. Simultaneously, the aluminum hydroxide flocculents can also adsorb marine larvae, further enhancing the antifouling effect.
Taking a copper-aluminum composite anode as an example, the outer copper layer and the inner aluminum layer undergo simultaneous electrolytic reactions. Copper ions are responsible for antifouling, while aluminum ions are responsible for corrosion prevention. The reaction current for both is uniformly regulated by the control cabinet to ensure that the copper ion concentration is maintained at the optimal antifouling level of 2 ppb. The aluminum hydroxide protective film stably adheres to the inner wall of the pipe. The spiral bifunctional anode with a built-in iron cathode also provides local cathodic protection to surrounding pipes through a cathodic reduction reaction, further reducing the corrosion rate of the pipes.
Applications of MGPS Anodes
The seawater systems of large cruise ships are complex, containing multiple branch pipelines and equipment. The installation location of MGPS anodes must adhere to the principles of “source protection, comprehensive coverage, and ease of maintenance” to ensure uniform ion protection from the seawater inlet to the end of the pipeline.
(I) Seawater Intake Grilles and Subsea Valve Boxes
The seawater intake grilles and subsea valve boxes are the “inlets” of the cruise ship’s seawater system. Marine larvae in the seawater first enter the pipeline through these points, making them the core source location for anode installation. Typically, 2-4 subsea valve boxes are installed on both the port and starboard sides of the cruise ship. Each valve box contains 2-3 sets of copper anodes or copper-aluminum composite anodes. Some large valve boxes are equipped with one set of iron anodes (for corrosion protection of the copper-nickel alloy valve box body).
The anode should be kept 10-15cm away from the inner wall of the valve box to prevent the accumulation of anode dissolution products at the bottom of the valve box. It should be fixed by welding or bolts to ensure an electrical contact resistance ≤0.01Ω to avoid affecting electrolysis efficiency due to poor contact. The anode position should avoid the water flow channel of the seawater intake grille to prevent affecting the seawater intake. The core function of the anode here is to inhibit the activity of marine organisms before they enter the pipeline through a high concentration of copper ions, reducing the risk of biofouling at the source.
(II) Seawater Filters Multiple
Seawater filters (including coarse and fine filters) are installed between the subsea valve box and the main pipeline to filter impurities in the seawater. These filters are also important intermediate nodes for anode installation. Each filter typically contains 1-2 sets of spiral bifunctional anodes or flanged composite anodes, with specifications selected according to the filter diameter (50mm-80mm diameter anodes for small filters, 100mm-120mm diameter anodes for large filters).
The anode is installed using a flange or sleeve type, directly fixed to the filter cover for easy disassembly and replacement during navigation. The anode must be aligned with the water flow outlet of the filter to ensure ions can quickly enter the main pipeline with the water flow. For PVC filters, a composite anode with a built-in iron cathode must be selected to form a complete electrolysis circuit. The core function of the anode here is to further increase the ion concentration in the seawater, kill any remaining marine larvae inside the filter, and protect the filter’s inner wall and subsequent pipelines from corrosion.
(III) Main Cooling Pipelines
The engine cooling system, air conditioning system, condenser, heat exchanger, and other core equipment on cruise ships are high-risk areas for biofouling and corrosion. At the inlet pipes of these devices, 2-4 sets of copper-aluminum composite anodes are typically installed; some high-temperature cooling pipes (copper-nickel material) will also have additional iron anodes installed.
The anode should be installed in a straight pipe section 1-2 meters before the equipment inlet to ensure that ions diffuse evenly in the water flow before entering the equipment. A safe distance must be maintained between the anode and the equipment, avoiding the equipment’s sensors and interfaces. For large equipment such as condensers and heat exchangers, small anodes can be installed on the tube sheet inside the equipment for direct internal protection. The core function of the anode here is to ensure unobstructed piping and heat exchange efficiency of the core equipment, preventing overheating and shutdown due to biofouling.
Related products
-
Impressed Current Cathodic Protection
ICCP MMO Rod Anode
-
Impressed Current Cathodic Protection
ICCP Cathodic Protection For Pipelines
-
Impressed Current Cathodic Protection
ICCP Cathodic Protection For Ships
-
Impressed Current Cathodic Protection
ICCP MMO Disk Anode
-
Impressed Current Cathodic Protection
ICCP MMO Tubular Anode
-
Impressed Current Cathodic Protection
Impressed Current Cathodic Protection System
-
Marine Growth Prevention Systems
MGPS Anode for Yachts
-
Marine Growth Prevention Systems
Aluminum Marine Growth Prevention Anode
























