Boats frequently navigate complex electrolytic environments such as coastal waters, estuaries, and harbors, where corrosion is particularly pronounced. The annual corrosion rate of the hull structure can reach 0.1-0.3 mm. Corrosion failure of critical components such as propellers and shafts is one of the main causes of navigation accidents. Zinc sacrificial anodes are the most mature, economical, and convenient solution for cathodic protection technology in ships. Through the principle of electrochemical sacrifice, they provide continuous and stable protection to the metal structure of the hull at the cost of their own preferential corrosion.
Types of Boat Zinc Anodes
Small boat zinc anodes are divided into various types based on differences in structural design, installation methods, and application scenarios. Different types have different focuses in terms of shape, installation, and protection range. The following focuses on five mainstream types:
Hull Zinc Anodes
Hull zinc anodes are primarily installed on the hull plating below the waterline, including critical areas of the bow, midship, and stern. They are especially suitable for the overall hull protection of steel and aluminum alloy small boats, and can also be used for corrosion protection of metal frames and fittings in wooden and fiberglass small boats.

- Rectangular Anodes
Weighing mostly 0.5-5kg, suitable for flat or gently curved areas of the hull.

- Square Anodes
Thinner in thickness, suitable for areas with limited space.

- Teardrop-shaped Anodes
Effectively reducing water flow impact and biofouling.
The material of ship hull zinc anodes is mainly zinc-aluminum alloy, including 0.3%-0.6% aluminum and 0.02%-0.07% cadmium. These elements improve current efficiency and corrosion uniformity. Its electrochemical performance is stable, with an open-circuit potential in seawater of -1.05V to -1.15V (relative to a saturated calomel electrode SCE). The working potential is -1.05V to -1.00V, providing a continuous cathodic protection current for the ship’s hull.
Bracelet Zinc Anode
The bracelet zinc anode (also known as a ring-shaped zinc anode) is a ring-shaped anode specifically designed for the shaft systems and propeller shafts of small boats. It is a core specialized anode for protecting shaft system components. Its structural feature is a hollow circular ring in the center. Its inner diameter precisely matches the outer diameter of the propeller shaft, with a thickness typically ranging from 20-50mm and a width of 30-80mm. The weight varies from 0.3-3kg depending on the shaft diameter.

The core advantage of the bracelet zinc anode lies in its precise protection of the critical area where the propeller shaft contacts seawater. The bracelet zinc anode also adheres to zinc-aluminum alloy standards. The pure zinc content is ≥99.9%, and the iron and copper content is ≤0.005%, ensuring a current efficiency of ≥95% in seawater and an actual electrical capacity of no less than 780 A·h/kg. It is suitable for the propeller shafts and drive shafts of small boats, especially for small motorboats with shaft diameters in the range of 20-100mm, such as speedboats, fishing boats, and sightseeing boats.
Welded Zinc Anodes
Welded zinc anodes are a type of anode fixed to the hull structure by welding. Their core characteristics are a secure connection and excellent conductivity, effectively preventing loosening or detachment due to ship vibration and water impact.
Welded zinc anodes typically have pre-set welding lugs or bases on the anode body. The welding lugs are made of a zinc alloy or low-carbon steel compatible with the anode material, ensuring that no brittle compounds are formed during welding. Welded zinc anodes come in various shapes, including block, plate, and strip forms. The weight ranges from 0.5 to 10 kg and can be customized according to the size and shape of the protected area.

Welded zinc anodes require a higher level of installation expertise. Before welding, oil, rust, paint, and other insulating layers must be removed from the anode surface and the welding area of the ship’s hull to ensure welding quality and electrical connection. After welding, the weld strength must be checked to avoid problems such as incomplete or faulty welds. The weld length is usually ≥50mm, and the height ≥3mm, to ensure connection stability. Welded zinc anodes are mainly used in areas of small vessels where the structure is relatively fixed and difficult to disassemble, such as hull strengthening plates, ballast tank bulkheads, rudder blades, and propeller hubs. Their advantage lies in high protective current transfer efficiency, with contact resistance ≤0.01Ω.
Bolt-on Zinc Anodes
Bolt-on zinc anodes are a type of anode installed using bolts, making them the preferred choice for routine maintenance and temporary corrosion protection of small boats. The core of their design is the anode body with pre-drilled bolt holes, typically 1-4 threaded holes of suitable diameter, along with matching stainless steel or galvanized bolts, nuts, flat washers, and spring washers.

The anodes are mainly block or plate-shaped, lightweight, and typically weigh 0.3-5 kg. Their surface is passivated. Some bolt-on zinc anodes also feature conductive gaskets at the contact surface between the anode and the hull to further reduce contact resistance and improve current transfer efficiency. In terms of material, bolt-on zinc anodes are the same as welded zinc anodes, conforming to the MIL-DTL-18001L standard, with a zinc content of ≥99.99% and impurity content strictly controlled to below 0.05%. Bolt-on zinc anodes are used on parts of the ship such as the rudder stock, water tank hatches, deck metal supports, and outboard engine mounts.
Boat Zinc Anode Strip
Boat zinc anode strip is a long, flexible anode product. It is primarily used for corrosion protection in confined spaces, irregular curved surfaces, or large areas on small boats. Its width is typically 25-100mm, thickness 1-5mm, and weight (by length) 0.5-5kg/m. In terms of electrochemical performance, marine zinc anode strips have a current efficiency of ≥95% in seawater and ≥65% in soil, with an actual capacitance of no less than 780A·h/kg.

The core advantage of zinc anode strips lies in their adaptability to the complex internal structure of small boats, especially in confined spaces such as ballast tanks, freshwater tanks, and fuel tanks. Furthermore, zinc anode strips can also be used on irregular curved areas such as below the deck and on both sides of the keel, achieving full coverage through flexible bonding.
Why Zinc Anodes?
The core purpose of using zinc anodes on boats is to inhibit the electrochemical corrosion of the hull’s metal structure through cathodic protection principles, ensuring safe navigation. To understand this necessity, a thorough analysis is required from three aspects: the nature of ship corrosion, the protection principle of zinc anodes, and their practical application value:
The Nature of Corrosion
During sailing and mooring, a boat’s underwater metal structures (such as the hull, propeller, shaft system, rudder blades, etc.) are always in an electrolyte environment (seawater, freshwater, soil, etc.). The structural materials of small boats include steel, aluminum alloys, copper alloys, and cast iron. Different materials have significantly different electrode potentials. When they are in contact and in the same electrolyte environment, galvanic corrosion inevitably occurs.
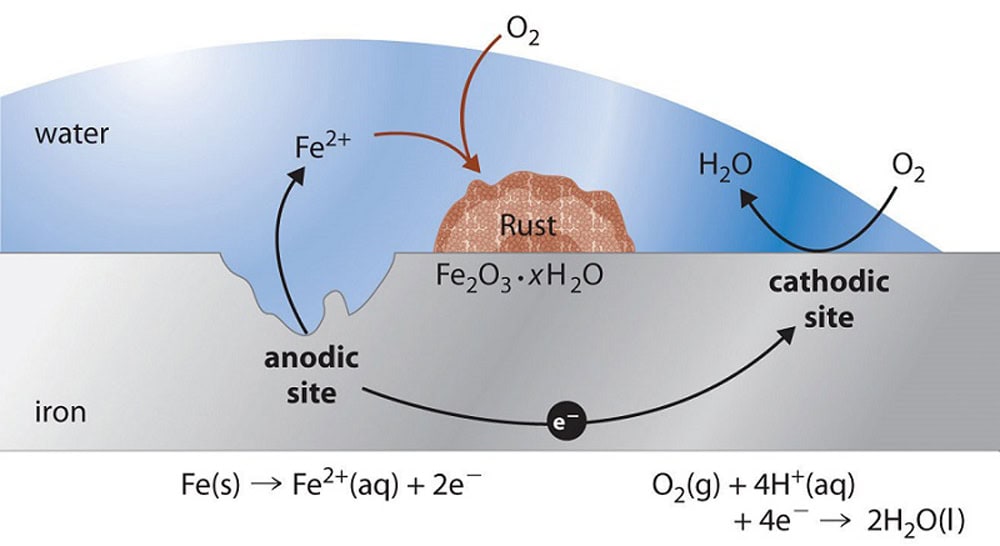
The essence of electrochemical corrosion is the formation of a galvanic cell on the metal surface, resulting in oxidation-reduction reactions: the metal loses electrons and is oxidized into ions, dissolving into the electrolyte, leading to the gradual deterioration of the metal structure. Data shows that the service life of underwater structures of steel boats without zinc anodes is typically 5-8 years. With zinc anode protection, the service life can be extended to 15-20 years or more.
Zinc Anode Cathodic Protection
The function of zinc anodes is based on “sacrificial anode cathodic protection.” The core mechanism is a redox reaction driven by a potential difference, making the protected hull metal the cathode and preventing its corrosion. The standard electrode potential of zinc is -1.10V (relative to a saturated copper sulfate reference electrode, CSE), while the electrode potential of steel commonly used in small boats is -0.76V (CSE). When the zinc anode is connected to the steel structure of the hull via a wire or direct contact, a spontaneous galvanic cell system is formed.
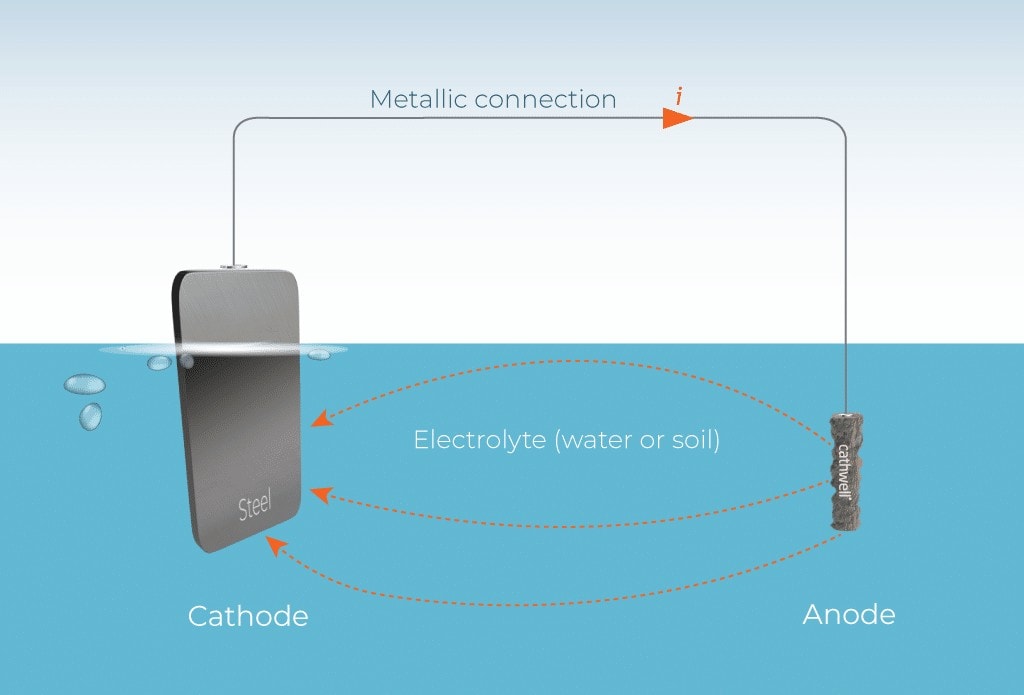
Redox Reaction: In this galvanic cell system, the zinc anode undergoes oxidation (corrosion and dissolution). Zinc atoms lose electrons and become zinc ions, entering the electrolyte: Zn → Zn²⁺ + 2e⁻; while the steel structure of the hull receives the electrons released by the zinc anode, inhibiting its own oxidation reaction (Fe → Fe²⁺ + 2e⁻), thus preventing the steel from corroding. The zinc anode “sacrifices” itself to protect the hull structure, hence the name “sacrificial anode.”
Corrosion Protection Effect
Zinc anodes come in various specifications (from small 0.3kg bolt-on anodes to 10kg block-type welded anodes), and can be installed in flexible ways (welding, bolting, adhesive bonding, etc.), adapting to different types and structures of small boats. Zinc anodes effectively inhibit various types of corrosion, including uniform corrosion, pitting corrosion, crevice corrosion, and galvanic corrosion, achieving a protection rate of over 90%.
Zinc Anode Replacement
The replacement cycle for zinc anodes on small boats is not a fixed value, but is influenced by various factors such as the operating environment, anode specifications, installation location, and hull material.
Environment
Environmental corrosiveness is a key factor affecting the consumption rate of zinc anodes. Small boats operating in seawater for extended periods experience high chloride ion concentrations, resulting in a faster anode consumption rate and a shorter replacement cycle. Small boats operating in freshwater have a higher electrolyte resistivity, leading to a slower anode consumption rate and a longer replacement cycle. Additionally, high-temperature seawater (water temperature > 35°C) can increase the anode corrosion rate by 30%-50%.
Specifications and Material
The weight, size, and material of the anode directly affect its lifespan. Larger anodes theoretically have a longer lifespan; for example, a 1kg zinc anode has a theoretical lifespan of approximately 1 year in seawater, while a 5kg zinc anode can last 3-5 years. Zn-Al-Cd anodes have a higher current efficiency than pure zinc anodes, corrode more evenly, and have a lifespan 20%-30% longer than pure zinc anodes.
Installation Location
Anodes on high-speed moving parts such as propeller shafts and rudders have a 30%-50% faster consumption rate than hull anodes.
Seawater Environment (Long-term sailing)
- Hull Anodes: 1-2 years
- Propeller Shaft Bracelet Anodes: 6-12 months
- Rudder Anodes: 8-15 months
- Ballast Tank Anode Strips: 3-5 years
Freshwater Environment (Long-term sailing)
- Hull Anodes: 2-4 years
- Propeller Shaft Bracelet Anodes: 1-2 years
- Rudder Anodes: 1.5-3 years
- Ballast Tank Anode Strips: 5-8 years
Mixed Environment (Alternating Seawater + Freshwater)
- Hull Anodes: 1.5-3 years
- Propeller Shaft Bracelet Anodes: 8-18 months
- Rudder Anodes: 1-2.5 years
- Ballast Tank Anode Strips: 4-6 years
It should be noted that the above reference ranges are theoretical estimates, and the actual replacement cycle needs to be adjusted based on regular inspection results. For example, for small boats that are often moored in ports and rarely sail, the anode consumption rate is slower, and the replacement cycle can be extended by 30%-50%; while for small boats that frequently sail in high-temperature, high-salinity waters, the replacement cycle needs to be shortened by 20%-40%.
Specifications of Zinc Anodes
The specifications of zinc anodes for small boats directly determine their protective effect. Strict adherence to international authoritative standards ensures that material purity, electrochemical performance, physical dimensions, and other indicators meet the requirements. The following details the specifications of zinc anodes for small boats from four core dimensions: zinc purity, standard specifications, physical dimensions, and electrochemical performance:
Zinc Purity
The purity of the zinc anode is a key factor affecting its electrochemical performance. According to ASTM F1182-07r19, “Standard Specification for Sacrificial Zinc Alloy Anodes,” the purity and impurity content requirements for small boat zinc anodes are as follows:
Zinc (Zn): As the base element of the anode, the content must be ≥99.9% (balance), ensuring the core electrochemical activity of the anode.
Aluminum (Al): As an alloying element, the content needs to be controlled between 0.3% and 0.6%. Aluminum can improve the current efficiency and corrosion resistance of the anode, forming a dense aluminum oxide protective film on the anode surface, slowing down the corrosion rate. However, excessive content will lead to anode passivation and affect current output.
Cadmium (Cd): As an alloying element, the content must be controlled between 0.02% and 0.07% (permission from the buyer is required before use). Cadmium can lower the corrosion potential of the anode, increase the driving voltage, improve corrosion uniformity, and avoid localized rapid consumption. However, cadmium is toxic, and its content must be strictly controlled to meet environmental requirements.
Impurity elements (such as iron, copper, lead, silicon, etc.) will seriously affect the electrochemical performance of the zinc anode, leading to reduced current efficiency, accelerated corrosion rate, and unstable potential. Therefore, their content must be strictly limited:
- Iron (Fe): ≤0.005%.
- Copper (Cu): ≤0.005%.
- Lead (Pb): ≤0.006%.
- Silicon (Si): ≤0.125%.
- Total impurity content: ≤0.1%.
Standards and Specifications
The manufacturing, quality inspection, and use of zinc anodes for small boats must comply with internationally recognized standards. Currently, the main standards and specifications include:
ASTM F1182-07r19
This standard, developed by ASTM International, is a widely recognized standard for zinc anodes in the international shipbuilding industry, applicable to cathodic protection of steel structures such as ships, submarines, and offshore platforms.
It covers various forms of zinc anodes, including plate, block, disc, and rod shapes, and is suitable for corrosion protection of hulls, submarine shells, seawater cooling systems, heat exchangers, and other components. Zinc anodes are divided into two classes: Class 1 for cored anodes (such as hull anodes with steel or brass cores), and Class 2 for coreless anodes (such as extruded rods and rolled plates).
Chemical composition: Zinc content ≥99.9%, aluminum content 0.3%~0.6%, cadmium content 0.02%~0.07%, and the content limits of impurity elements such as iron, copper, and lead are basically consistent with GB/T 4950-2021.
Electrochemical performance: Open circuit potential -1.05V~-1.15V (SCE), working potential -1.00V~-1.05V (SCE), current efficiency ≥95% (in seawater), actual capacity ≥780 A·h/kg.
Military Standard: MIL-A-18001K
This standard is the governing standard for zinc anodes used in US military vessels, requiring stricter specifications and applicable to military ships and other vessels. Zinc content ≥99.95%, impurity elements: iron ≤0.003%, copper ≤0.002%. It offers stable electrochemical performance, longer service life, and reliable protection in harsh environments.
- Open Circuit Potential
- Environment: 25℃ artificial seawater (sodium chloride content 3.5%),
- Reference electrode: Saturated calomel electrode (SCE),
- Standard value: -1.05V~-1.15V,
The open circuit potential reflects the electrochemical activity of the anode. A potential that is too positive will result in insufficient driving voltage and poor protection; a potential that is too negative may cause overprotection, leading to hydrogen embrittlement of the hull.
- Current Efficiency
- Test conditions: In seawater,
- Standard value: ≥95% (in seawater), ≥65% (in soil),
Current efficiency is the ratio of the actual amount of electricity released by the anode to the theoretical amount, reflecting the energy utilization rate of the anode. The higher the current efficiency, the less the anode itself is consumed, and the more durable the protection effect. For example, a current efficiency of 95% means that 95% of the electricity released by the anode is used to protect the hull, and only 5% is consumed by ineffective self-corrosion.
- Driving Voltage
- Environment: Seawater
- Standard Value: ≥0.25V
The driving voltage is the potential difference of the galvanic cell formed between the anode and the ship’s hull, ensuring sufficient current output. Insufficient driving voltage will result in insufficient protective current, which cannot effectively inhibit hull corrosion.
Wstitanium's Advantages
Wstitanium is a company specializing in the research, development, and manufacturing of high-end sacrificial anodes. Its small boat zinc anodes possess significant advantages in the industry due to their advanced material formulation, precise manufacturing, and strict quality control.
Optimized Material Formulation
The core advantage of Wstitanium zinc anodes lies in their unique zinc alloy formula. Based on the ASTM F1182-07r19 standard, the precise control of alloy element ratios and strict control of impurity content have resulted in a comprehensive improvement in electrochemical performance.
Wstitanium zinc anodes have a zinc content of up to 99.95%. The content of impurity elements iron, copper, and lead are controlled to below 0.002%, 0.001%, and 0.003%, respectively, with a total impurity content of ≤0.05%. For example, a 1kg Wstitanium hull zinc anode has an actual electrical capacity of 820 A·h/kg, providing a theoretical protection capacity of 820 A·h in seawater, while ordinary zinc anodes (780 A·h/kg) have a theoretical protection capacity of 780 A·h. The former has a 5.1% longer service life than the latter.
Manufacturing
Wstitanium employs internationally advanced manufacturing technologies and equipment, establishing a full-process quality control system to ensure that every batch of zinc anodes meets high-end standards. Wstitanium zinc anodes are manufactured using precision casting. The internal structure of the anode is denser, with a density of 7.1 g/cm³ (higher than the standard requirement of 6.8 g/cm³).
Quality Inspection
Chemical composition: Using a spectrometer, 100% testing is performed on the content of zinc, aluminum, cadmium, and impurity elements to ensure compliance with formula requirements;
Electrochemical performance: Samples are randomly selected to test the open-circuit potential, working potential, actual capacity, and current efficiency to ensure compliance with internal corporate standards (5% higher than national standards);
Physical properties: Dimensional accuracy, surface quality, density, and mechanical strength are tested, and unqualified products are rejected;
Corrosion: A 1000-hour accelerated corrosion test is conducted to observe the uniformity of corrosion and the protective effect, ensuring that the service life meets the standards.
Cathodic Protection Effectiveness
Compared to ordinary zinc anodes, Wstitanium zinc anodes offer significant advantages in terms of protection efficiency, service life, and environmental adaptability, providing more comprehensive and longer-lasting protection for small boats. Wstitanium zinc anodes have a current efficiency of over 97%, improving protection efficiency by 10%-15% compared to ordinary zinc anodes. For example, small boats equipped with Wstitanium zinc anodes can reduce hull corrosion rates to below 0.015 mm/a, achieving a protection rate of over 95%.
Seawater environment: The service life of 1 kg of Wstitanium hull zinc anode is approximately 1.2-1.3 years, while that of an ordinary zinc anode is approximately 1 year;
Freshwater environment: The service life of 1 kg of Wstitanium hull zinc anode is approximately 2.4-2.6 years, while that of an ordinary zinc anode is approximately 2 years.
Customization
Wstitanium offers a wide range of customizable specifications, tailoring zinc anodes to the specific material (steel, aluminum alloy, wood, fiberglass), size, and structural characteristics of the boat. For example: for aluminum alloy boats, we customize low-potential zinc anodes (open-circuit potential -1.05V~-1.08V) to prevent excessive potential difference between the anode and the hull, thus avoiding accelerated hull corrosion;
Environmental Protection
Wstitanium zinc anodes strictly adhere to international environmental standards and comply with international environmental directives such as RoHS and REACH. The cadmium content of our zinc anodes is controlled at 0.04% to 0.06%, meeting the requirements of ASTM F1182-07r19 standard (“requires customer approval before use”). We also offer a cadmium-free formula (adding 0.5% to 0.8% aluminum as a substitute for cadmium) to meet the needs of environmentally sensitive areas.
Conclusion
Boat zinc anodes are a core material for cathodic protection. Based on electrochemical principles, zinc anodes provide a continuous protective current to the underwater metal structures of the boat through preferential corrosion, inhibiting electrochemical corrosion and serving as an essential means of corrosion prevention for small boats. Their protection rate can reach over 90%. Small boat zinc anodes mainly include five types: hull, bracelet, welded, bolted, and zinc strip anodes. The appropriate type should be selected and installed based on the corrosion risk and structural characteristics of different parts (hull, propeller shaft, rudder blade, ballast tank, etc.). Small boat zinc anodes must comply with authoritative standards such as ASTM F1182-07r19, with a zinc purity of ≥99.9%, current efficiency of ≥95% (in seawater), actual capacity of ≥780 A·h/kg, and dimensional tolerance of ≤±3mm, ensuring stable electrochemical performance.
















